
It shows the product of uncertainties in position and momentum is of order of Plank’s constant. Implementations of joint measurements of position and momentum and commentīriefly on the status of experimental tests of the uncertainty principle.A particle is considered as a wave packets. 
No According to the Heisenberg Uncertainty Principle, it is impossible to simultaneously determine the and the of a particle. Finally, we survey models and experimental According to Heisenbergs uncertainty principle: The position and momentum of a small particle cannot be determined simultaneously The Heisenberg Uncertainty Principle is valid at the macroscopic level. WhilstĬonceptually distinct, these three kinds of uncertainty relations are shown toīe closely related formally. Quantities and for the inaccuracy of a measurement of one of the quantitiesĪnd the ensuing disturbance in the distribution of the other quantity. According to the Heisenberg uncertainty principle, if the uncertainty in the speed of an electron is 3.5 x 10 m/s, the uncertainty in its position (in m) is at least A. Quantum state for the inaccuracies of any joint measurement of these The uncertainty principle, according to which the uncertainty in energy of a level is inversely proportional to the lifetime, tells us that T2 is a measure. Expert Answer 100 (1 rating) Transcribed image text: 6. Relations: for the widths of the position and momentum distributions in any According to Heisenbergs uncertainty principle, the product of uncertainties in position and velocities for an electron of mass 9.1 x 10-31 kg is (1) 2.8 x 10-mas-1 (2) 3.8 10- ms-1 (3) 5.8 x 10-5 ms-1 (4) 68 x 10ms 19. it is one of the most famous (and probably misunderstood) ideas in Physics. The uncertainty principle says that it is not possible to precisely and simultaneously measure both the velocity and position of a subatomic particle like an electron. Is shown to appear in three manifestations, in the form of uncertainty Heisenberg’s Uncertainty principle is one of the most important tenets of this field. Thus, we did not extend Bohr’s model to other atoms because of these limitations. According to the uncertainty principle the particle at rest is everywhere in space simultaneously, as you say. On the other hand there are no infinities. Therefore, Bohr’s model of the hydrogen atom not only ignores the dual behaviour of matter but also contradicts Heisenberg’s uncertainty principle. The bound electron fulfills the Heisenberg uncertainty principle (HUP) as it is expressed as a solution of Schroedingers equation. Reconciled if an appropriate trade-off is accepted. According to Heisenberg’s uncertainty principle, this is not possible. That the full content of this principle also includes its positive role as aĬondition ensuring that mutually exclusive experimental options can be The Heisenberg Uncertainty Principle states that you can never simultaneously know the exact position and the exact speed of an object. Of operational possibilities imposed by quantum mechanics. 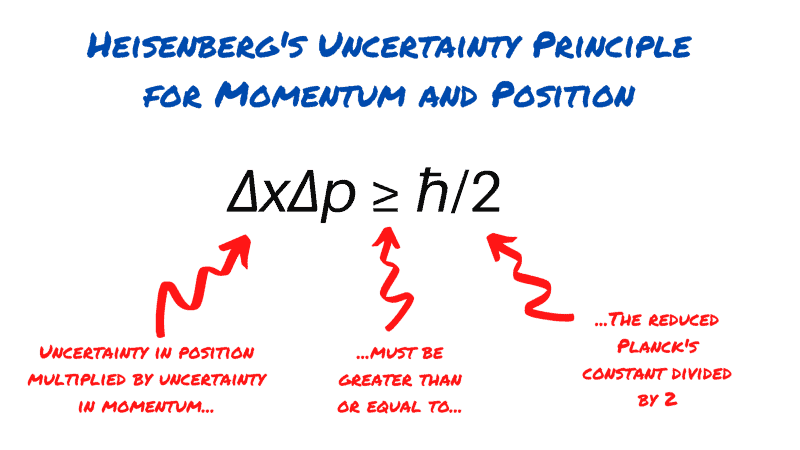
Busch and 2 other authors Download PDF Abstract: Heisenberg's uncertainty principle is usually taken to express a limitation Download a PDF of the paper titled Heisenberg's Uncertainty Principle, by P. HEISENBERG’S UNCERTAINTY PRINCIPLE PAUL BUSCH, TEIKO HEINONEN, AND PEKKA LAHTI Abstract.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
